
Emission spectra are discontinuous atomic spectra as the light is emitted only at a certain wavelength with dark spaces between them and absorption spectra are continuous atomic spectra as a continuum of radiation is passed through a sample which absorbs radiation of specific wavelength. They emit radiation over a broad range of wavelengths thus the spectra appear smooth and continuous. It arises from dense gases or solid objects which radiate heat. Note: Continuous spectra also called thermal or blackbody spectra. The main difference is that line spectra can be seen as either isolated emission lines or absorption lines, with huge gaps between them, whereas continuous spectra do not contain gaps and can be produced by superimposing the emission and absorption spectra of the same. There is nothing between each line, so the spectrum is discontinuous. Each packet of energy corresponds to a line in the atomic spectrum. Hence, atomic emission spectra represent the electrons returning to lower energy levels. This release occurs in the form of light of a specific wavelength (colour). When it returns to a lower level, it releases energy in a quantized packet. If an electron is excited to a new energy level, it jumps to that level instantaneously. This is called a continuous emission spectrum. white light through a gas certain colors of light are absorbed by the gas, causing black bars to appear. , shows all wavelengths of visible light. When an absorption spectrum and emission spectrum are combined, you get a continuous spectrum with all the wavelengths. The sun’s light is also believed to produce continuous spectrum since we can see the rainbow. This light, when seen through a prism or diffraction grating. Truly white light, like that emitted by some stars and moons is a perfect example of continuous spectrum. The electrons in an atom can have only certain energy levels. When an absorption spectrum and emission spectrum are combined, you get a continuous spectrum with all the wavelengths. Atomic spectra are discontinuous because the energy levels of electrons in atoms are quantized. The continuum, is NOT any comb of densely packed line spectra, it is a truly continuous spectrum, with any wavelength or frequency beyond the atomic line spectrum limit. Each of these mechanisms has a characteristic spectrum.  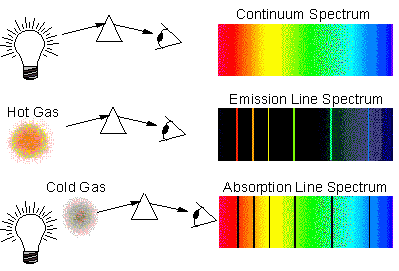
For instance, there are many different mechanisms by which an object, like a star, can produce light. Each spectrum holds a wide variety of information. A discontinuous spectrum is a spectrum that contains gaps, holes, or breaks in terms of the wavelengths that it contains.Ītomic spectra also known as line spectra. Spectra can be produced for any energy of light, from low-energy radio waves to very high-energy gamma rays. A continuous spectrum contains every wavelength between the wavelength on which the spectrum starts and the wavelength on which the spectrum ends. Electromagnetic spectrums classify into continuous spectrum and discontinuous spectra. To understand that the spectral emission lines of an element have the same frequencies as the lines of the absorption spectrum.Hint: We now know that atoms are made up of protons, neutrons and electrons.To characterize an atom by its light spectrum and to introduce spectroscopy.To distinguish between absorption spectra and emission spectra.CRC Handbook of Chemistry and Physics NSRDS-NBS 68 (1980).Ĭlick on an element to select it. Source for the values of spectral lines: CDS Strasbourg University ( link) from Reader J., and Corliss Ch.H. For this reason, a gas composed of a single atom can absorb or emit a limited number of frequencies.įor a given element, the emission spectrum (upper part of the animation) has the same frequency as its absorption spectrum (bottom part). It is "quantized" (see animation line spectrum of the hydrogen atom). Each thin band in each spectrum corresponds to a single, unique transition between energy levels in an atom. 5: The atomic emission spectra for various elements. One of the great discoveries of quantum mechanics is that the energy of an atom can only have certain well-defined values. Scientists needed a fundamental change in their way of thinking about the electronic structure of atoms to advance beyond the Bohr model. For example, hydrogen has one electron, but its emission spectrum shows many lines. The amount the standard absorbs is compared with the. The number of lines does not equal the number of electrons in an atom. The spectrum obtained can be continuous or discrete ("line spectrum"). continuous spectrum is viewed behind a cool gas under pressure. Explanation: Continuous spectrum: a spectrum that has all wavelengths with no gaps over a wide range. A prism (or an array) is used to break a beam of light according to its different frequencies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
